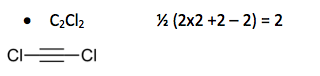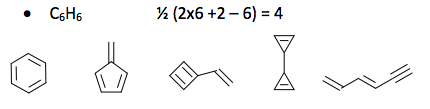How to Calculate the Number of Double Bonds or Rings in an Organic Structure
How to Calculate the Number of Double Bonds or Rings in an Organic Structure
Organic compounds are said to be saturated when they have no double bonds and unsaturated when they have double bonds. In fact the term polyunsaturated refers to long chain organic compounds with more than one double bond. The presence of double bonds is known as the degree of hydrogen deficiency (IHD) or degree of unsaturation (DOU). The IHD also applies to organic ring compounds. It is helpful to know the IHD, when determining compound structures. It is a useful tool when solving spectroscopy problems. The IHD can be calculated by the formula: ½(2C + 2 + N – H – X) where x = F, Cl, Br, I. Oxygen is not considered in the calculation.
Consider the following examples. Find the IHD and draw matching constitutional structures where possible. Constitutional structures are those with the same molecular formulas but different connectivity.

New Paragraph
New Paragraph
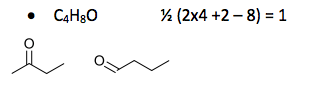
New Paragraph
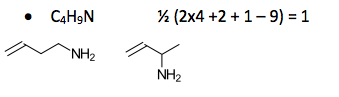
New Paragraph